Newsletter
Test Ordering Risks and Remedies
Sep 26, 2023
Identifying a prevalent patient safety risk only has value if we also understand how it might be mitigated.
Across the country, MPL insurers and those who deliver risk management services are being asked by providers to help them identify and implement solutions. To unravel the myriad effective practices, tools, strategies, and success stories, Candello (a division of CRICO) has created a Risk Interventions Library. This collection offers proven initiatives to address some of the most common contributing factors leading to medical error.
For example, patient assessment represents a primary factor contributing to MPL claims that often involve high clinical severity and significant incurred loss. More specifically, the risk of a failure or delay in diagnostic testing is a clinical vulnerability for which there are many proven interventions and support resources.
Clinical Severity Linked to Failure or Delay in Diagnostic Testing
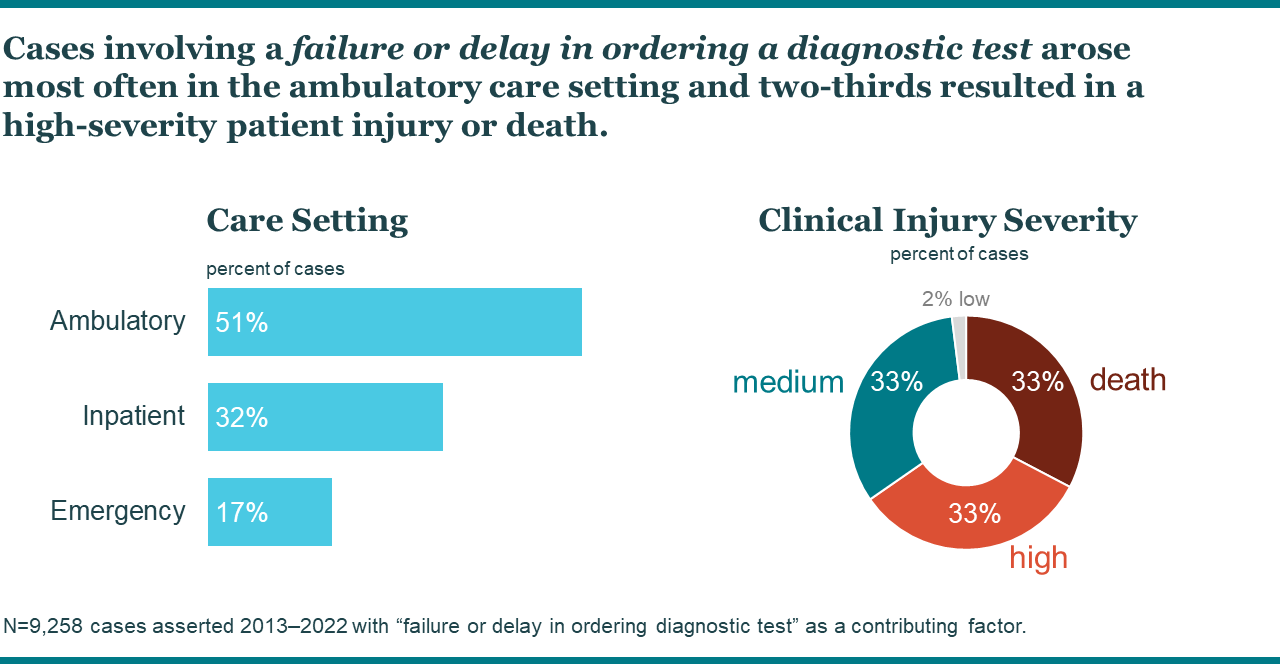
Ten years of data from the national Candello database indicate that a failure or delay in ordering a diagnostic test contributed to 14 percent of cases and 24 percent of losses.* Not surprisingly, cases involving this factor arose most often in ambulatory care. However, it was a frequent factor in all care settings. More striking is that two-thirds of patients experienced a high-severity patient injury or death when this factor was present.
*N=66,000 MPL cases asserted 2013–2022 with $17B total incurred losses.
To help insurers and health care leaders address this leading vulnerability for those delivering front-line care, Candello developed a list of decision support guides and interventions. If any of these solutions can be leveraged to address your needs, or you have a proven solution not listed here, please contact the team to share your experience.
Decision Support Guides
-
Appropriate Use Criteria (AUC): Initially developed for use in determining the appropriate use of imaging and diagnostic testing for Medicare patients, other examples of AUCs have been developed to rate the appropriate use of testing under various conditions. These scoring systems can be integrated into clinical decision support systems.
-
SAFER Guide for Test Results Reporting and Follow-up: The SAFER guide identifies recommended safety practices intended to optimize the safety and safe use of processes and EHR technology for the electronic communication and management of diagnostic test results.
-
Diagnostic Algorithms: The Mayo Clinic and the American Academy of Family Physicians publish lists of diagnostic algorithms for a wide range of conditions and situations. These algorithms provide guidance to aid in clinical decision-making with regard to ordering and managing diagnostic testing.
-
Electronic trigger-based interventions: A study in the Journal of Clinical Oncology found that an electronic trigger-based system can help to identify patients who may be at risk of diagnostic delays due to lack of follow-up and diagnostic testing.
-
Laboratory Test Catalogs: Mayo Clinic Laboratories also has an inventory of test catalogs that provide detailed information on laboratory tests available at a facility, along with guidance to help clinicians select appropriate tests for specific conditions.
Interventions
-
A systematic review published in 2019 evaluated the effectiveness of audit and communication strategies to reduce diagnostic errors made by clinicians. They found some evidence that trigger algorithms, additional patient reviews, team meetings, and error documentation may reduce diagnostic errors in different settings.
-
Starting in 2021, CRICO began the development and implementation of Ambulatory Safety Nets (ASNs) for top-occurring cancers and selected behavioral health issues. Forming a robust collaborative of members across the Harvard medical system, patients who previously fell through the cracks after a concerning clinical finding related to breast, colorectal, prostate, or lung cancer will now be systematically identified for follow-up treatment and closure.
-
The American Society for Clinical Laboratory Science published an article on how laboratory professionals can help to reduce diagnostic errors by providing feedback, reflexive testing, consultative services, and improved test reporting.
-
The Patient Safety Network provides information on clinical decision support systems (CDSS) that can help prevent prescribing errors, improve diagnostic accuracy, and enhance patient safety.
-
The Office of the National Coordinator for Health Information Technology publishes the SAFER Guide, which includes a self-assessment tool for evaluating and improving current practices around management and follow-up of diagnostic test results.
-
The Office of the National Coordinator for Health Information Technology (ONC) highlights how health records can improve the ability to diagnose diseases and reduce or prevent medical errors.
References
Abimanyi-Ochom J, Bohingamu Mudiyanselage S, Catchpool M, Firipis M, Wanni Arachchige Dona S, Watts JJ. Strategies to reduce diagnostic errors: a systematic review. BMC Medical Informatics and Decision Making. 2019;19(1):174. doi:10.1186/s12911-019-0901-1
Moyal-Smith R, Margo J, Maloney FL, Boulanger J, Lautner P, Folcarelli P, Benjamin E. The patient safety adoption framework: A practical framework to bridge the know-do gap. Journal of Patient Safety. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
Murphy DR, Wu L, Thomas EJ, Forjuoh SN, Meyer AN, Singh H. Electronic trigger-based intervention to reduce delays in diagnostic evaluation for cancer: A cluster randomized controlled trial. Journal of Clinical Oncology. 2015;33(31):3560-3567. doi:10.1200/JCO.2015.61.1301
Murphy DR, Meyer AN, Sittig DF, Meeks DW, Thomas EJ, Singh H. Application of electronic trigger tools to identify targets for improving diagnostic safety. BMJ Quality & Safety. 2019;28(2):151-159. doi:10.1136/bmjqs-2018-008086
Palestro CJ, Abikhzer G, Bar-Sever Z, Bartel T, Brady R, Grady EE, Israel O, Jain SK, Kandiah S, Sathekge MM, Shulkin BL. Appropriate use criteria for the use of nuclear medicine in fever of unknown origin. Society of Nuclear Medicine and Molecular Imaging. 2023. https://www.snmmi.org/ClinicalPractice/content.aspx?ItemNumber=43480
Schiff GD, Hasan O, Kim S, Abrams R, Cosby K, Lambert BL, Elstein AS, Hasler S, Kabongo ML, Krosnjar N, Odwazny R, Wisniewski MF, McNutt RA. Diagnostic error in medicine: Analysis of 583 physician-reported errors. Archives of Internal Medicine. 2009;169(20):1881-1887. doi:10.1001/archinternmed.2009.333
Recent Issues
Safety Nets for Safer Care


Recently Asked Questions

